Constraints
Constraints derived from over 250 experimental findings correlated with behaviors indicative of the generation of first-person properties are examined. Of these, nearly 50 were used to systematically restrict the space of a unique viable solution capable of generating both behavior and first-person properties. It is expected that constraints from the remaining findings will reveal features enabling seamless interconnections.
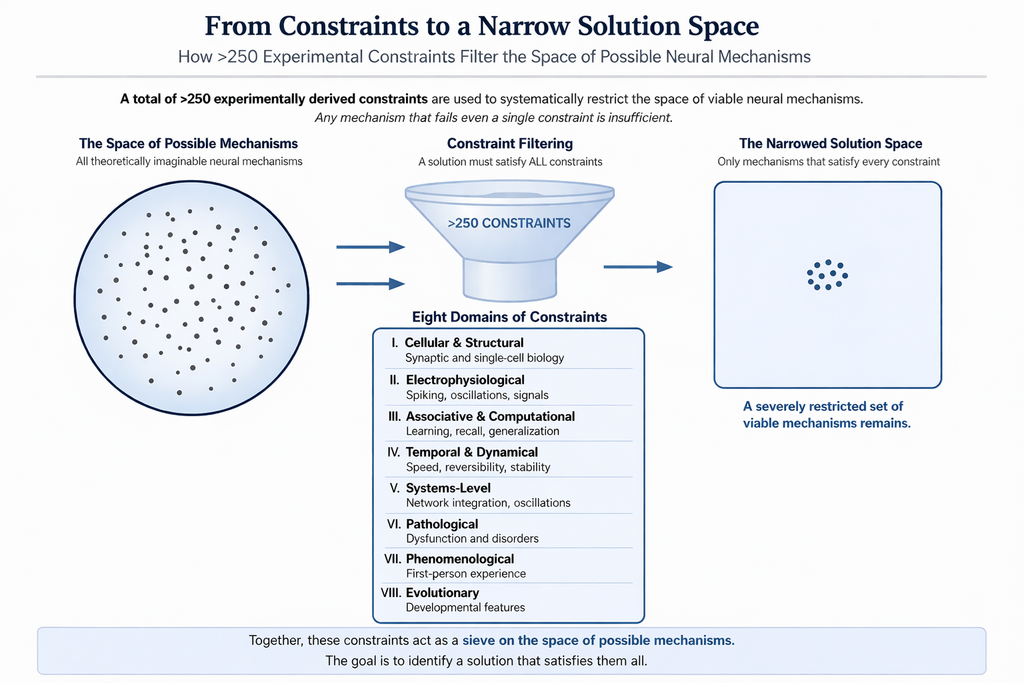
How hidden is the solution & how do the constraints help to find it?
The disparate findings from eight domains of the system functions (given after the next figure) are expected to be interconnected through a novel mechanism (Fig.2), analogous to an adaptor protein that binds to multiple molecular partners. This explains how the unique properties of the solution enable large number of features of the system observed from different levels to get integrated in an interconnected manner.
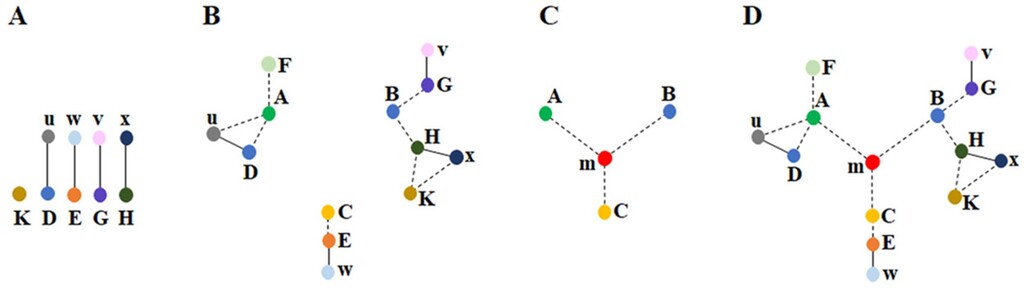
Figure 2. Disparate findings of the system in different levels are interconnected with each other through a unique solution A) The features of the system are detected either directly (represented by the capital letter K) through our sensory systems or indirectly (represented by the capital letters D, E, G, H) through findings such as protein staining or behavioral observations. These features are connected through smaller letters (u, w, v, x), which represent relationships between the features (e.g., the observation of u enables the detection of D). B) By utilizing both commonly employed direct and indirect methods, three clusters of interconnected findings (depicted by dotted lines) are identified across different levels (observations from various fields of brain science). In many instances, it is not possible to establish connections between these clusters. For example, no direct connection was found between: 1) the changes in learning and the internal sensations of memory occurring at millisecond timescales, and 2) sleep & long-term potentiation (LTP). However, by examining the constraints within each cluster, we can assess whether they can be unified through a common operational mechanism. In the context of the nervous system, a vast array of findings and the constraints they provide can be explored. C) By leveraging the constraints from certain features within each cluster of seemingly unrelated findings (e.g. A, B, C), it may become possible to derive a deep underlying principle (a structure-function solution m) that enables their interconnection. This solution is expected to offer a mechanism for generating internal sensations at millisecond timescales. D) The solution m provides an explanation of how various findings within each cluster are interrelated with one another & with findings from other clusters, as shown in B). The ability of solution m to integrate findings across all clusters makes it a further verifiable solution (Fig. from Vadakkan KI (2019) Phys Life Rev.31:44-78.
Constraints (~80) that were used to derive and verify the solution at the initial stage | |
| Finding | Constraint (The solution must be able to provide interconnected explanations for all the constraints). |
| Domain 1 Cellular and Structural a) For associating two stimuli & generating motor output reminiscent of the 2nd stimulus, in response to the 1st stimulus | |
| All the stimuli during learning & memory retrieval propagate through synaptically-connected neuronal circuits. | The mechanism should be compatible with signal propagation through synaptically connected neuronal circuits. |
| Any subset of 140 input signals reaching the axonal hillock is sufficient to fire a neuron (Palmer et al., 2014; Eyal et al., 2018). | This extreme degeneracy suggests that information specificity is unlikely to be encoded solely at the level of neuronal firing and instead may depend on changes at the level of synaptic input terminals. |
| Spine neck resistance (Tønnesen et al., 2014) and attenuation of potentials as they propagate along the dendrite towards the soma (Spruston, 2008). | Electrical compartmentalization by spine neck resistance and dendritic attenuation suggests that learning-related changes are likely to occur locally at or near the spine head. |
| Optogenetic activation of presynaptic inputs in lateral amygdala (LA) forms associative fear memory (Kwon et al., 2014). | Formation of associative fear memory requires convergence of associatively related inputs within LA synaptic networks. |
| Fear learning generates local connectivity between lateral amygdala (LA) neurons (Abatis et al., 2024) as evidenced by depolarization in a small subset of neighboring LA neurons, when a single LA neuron is stimulated. | Fear learning is expected to induce functional lateral interactions between neighboring LA neurons, potentially mediated at the level of dendritic spines. |
| Since associatively learned inputs synapse on to the spines of LA neurons, the inter-neuronal interaction is expected to take place between the synapses. | Since associative inputs terminate on dendritic spines, the minimal site capable of mediating direct inter-neuronal functional interaction is likely to involve interactions between postsynaptic spines. |
| Synapse-dense cortical areas with tightly packed neuropil have adjacent spines (Kasthuri et al., 2015; Zhu et al., 2021; Gemin et al., 2021). | Close spatial proximity of spines increases the likelihood of inter-neuronal inter-spine interactions. |
| The dendritic arbors of pyramidal neurons display significant territorial overlap (Mizuseki et al., 2011; Bezaire & Soltesz, 2019; Iascone et al., 2020). | Territorial overlap between dendritic arbors increases opportunities for inter-neuronal inter-spine interactions. |
| The sister branches on a neuron’s dendritic tree often avoid overlapping (Grueber & Sagasti, 2010). | Reduced overlap between sister dendritic branches may favor closer apposition between dendrites belonging to different neurons. |
| The mean inter-spine distance on the dendrite of a pyramidal neuron exceeds the mean spine diameter (Konur et al., 2003). | Greater spacing between spines on the same dendrite may favor interactions between spines belonging to different neurons rather than between neighboring spines of the same neuron. |
| Over half of the spine surface area lacks ensheathment by astrocytic processes (Ventura & Harris, 1999). | The non-ensheathed portion of spine membrane remains available for potential interactions with adjacent spines belonging to other neurons. |
| Synapses devoid of astrocytic coverage emerge in the amygdala during the consolidation of Pavlovian threat conditioning (Ostroff et al., 2010). | Reduction of astrocytic coverage during memory consolidation may increase opportunities for direct inter-neuronal inter-spine interactions. |
| Most excitatory glutamatergic synapses are located on dendritic spines, which enlarge during learning. Glutamate induces spine enlargement in both hippocampal slices (95%) (Matsuzaki et al., 2004) & the neocortex in vivo (22%) (Noguchi et al.,2019). | Learning-associated spine enlargement can increase membrane surface area and locally displace extracellular matrix components, thereby increasing the probability of inter-neuronal inter-spine interactions. |
| Fear conditioning is associated with enlarged synapses on the dendritic spines of LA neurons (Ostroff et al., 2010; Choi et al., 2021). | Synaptic enlargement accompanying fear conditioning is compatible with learning-induced structural interactions between neighboring spines. |
| Motivation enhances learning & is associated with the release of dopamine, which activates dopamine receptors in various regions of the brain (lino et al., 2020). | Dopamine-induced spine enlargement (Yagishita et al., 2014) may increase the likelihood of interactions between adjacent spines belonging to different neurons. |
| Contextual fear conditioning recruits newly synthesized GluA1-containing AMPA receptors into the spines of hippocampal memory-ensemble cells in a learning-specific manner (Matsuo et al., 2008). | Recruitment of GluA1-containing AMPA receptors through vesicular exocytosis adds membrane material to dendritic spines, potentially facilitating inter-spine interactions. |
| Autophagy leads to memory destabilization & erasure of auditory fear memories, a process associated with AMPA receptor endocytosis (Shehata et al., 2018). | AMPA receptor endocytosis removes membrane material from dendritic spines and may reduce or reverse previously established inter-spine interactions. |
| An inhibitor of AMPA receptor endocytosis partially rescued long-term memory deficits in mice with elevated levels of amyloid-β (Yan et al., 2024). | Inhibition of AMPA receptor endocytosis may stabilize spine structure and help preserve previously established inter-spine interactions. |
Mice injected with histone acetyl transferase (HAT) exhibit enhanced fear memory. Neurons in which HAT was overexpressed are part of the engram (Santoni et al., 2024). Removal of histone protein from DNA enhanced fear memory. The study found that a) neurons in which HAT is over-expressed are the neurons that fire during memory retrieval, & b) optogenetic silencing of these specific set of neurons prevents fear memory recall. | Histone acetylation enhances gene expression that may support structural and functional modifications required for inter-spine interaction mechanisms. Enhanced plasticity in HAT-overexpressing neurons may increase the probability of forming stable learning-related inter-spine interactions. |
| Most learning events result in a working memory that lasts only for a short period. All long-term memories has had working memories immediately after learning. | The mechanism should support both rapidly reversible interactions underlying working memory and more stabilized interactions capable of supporting long-term memory. |
| The capacity to store extensive sets of learning-induced changes underlies the ability to retrieve a large number of behavioral expressions during memory retrieval events. Using a finite number of dendritic spines this must be achieved. | The mechanism should permit combinatorial organization of learning-induced changes, enabling a finite number of spines to support large memory-storage capacity. |
| Higher brain functions occur only when the frequency of oscillating extracellular potentials falls within a narrow range, as evidenced by EEG recordings (Rusalova, 2006). | Interactions occurring along different spatial orientations within cortical neuropil may contribute vector components to extracellular oscillatory fields. Synaptic transmission in one direction and possible propagation of potentials across the perpendicularly occurring inter-spine interactions can contribute vector components for oscillations. |
| Even though extracellular recordings like EEG is biased by filtering effects (e.g. extracellular tissue act as a low-pass filter), brain operates in a narrow range of frequencies of EEG waveforms recorded from specific locations. For e.g. alpha rhythm (8–12 Hz) recorded from occipital cortex during relaxed wakefulness (Niedermeyer & da Silva, 2017). | The operational mechanism should be capable of contributing to oscillatory extracellular potentials and associated intracellular ionic dynamics. |
| A disconnect between dendritic depolarization & neuronal firing has been observed during fear conditioning (d’Aquin et al., 2022). | The operational mechanism underlying associative learning may emerge at the dendritic level independently of action potential generation. |
| b) For generating first-person property reminiscent of 2nd stimulus, in response to the 1st stimulus | |
| Memories are virtual first-person inner sensations. | It is necessary to identify the neural substrate and operational mechanism responsible for generating internally experienced memories. |
| Memory retrieval occurs on a millisecond timescale. | Since memories can be retrieved immediately after learning, the operational mechanism must function within milliseconds. |
| After associative learning between two items, the presentation of one item triggers the memory of the other. | The mechanism must explain how either member of an associative pair can function as a cue for retrieval of the other, implying bidirectional associativity. |
| A narrow range of oscillating extracellular potential frequencies is necessary to maintain normal consciousness during which learning and memory retrieval take place. | The mechanism underlying learning and memory retrieval must operate in a manner compatible with physiologically maintained oscillatory brain activity associated with consciousness. |
| Gradual changes in the qualia of the inner sensation of memory occur as the cue stimulus changes progressively. | The mechanism should support dynamic integration of elementary operations capable of generating continuously varying inner experiences. |
| Absence of cellular changes during memory retrieval. | Memory retrieval should occur through reactivation of learning-induced changes without requiring the formation of new cellular modifications during retrieval. |
| Instant access to very large memory stores. | The mechanism must explain how large memory stores can be accessed rapidly and efficiently through parallel or combinatorial operations. |
The inner experience of certain higher brain functions can occur without any accompanying motor actions. | The mechanism underlying inner sensations must remain functional even in the absence or suppression of behavioral motor actions. |
| Domain 2 Electrophysiological a) To explain Long-term potentiation (LTP), an electrophysiological correlate of the ability to learn | |
| Strength of LTP induced is correlated with the ability to learn (Matynia et al., 2002; Lynch MA, 2004). | The mechanisms underlying learning and LTP should share common cellular or synaptic processes. |
| Dopamine enhances both motivation-driven learning (Bromberg-Martin et al., 2010) & LTP (Otmakhova & Lisman, 1996). | Dopamine is known to cause increase in the size of spines (Yagishita et al., 2014). Dopaminergic modulation should influence structural or functional synaptic changes associated with both learning and LTP. |
| An increase in the amplitude of miniature EPSPs (mEPSPs) occurs following LTP induction (Manabe et al., 1992). Amplitude of mEPSPs is thought to be influenced by an increase in the number or functional efficacy of AMPA receptors (Malenka & Nicoll, 1999). | The mechanism underlying LTP should explain the observed increase in mEPSP amplitude following LTP induction. |
| Learning can be occluded following LTP induction, & conversely, LTP can be occluded after learning (Moser et al., 1998; Whitlock et al., 2006). | Learning and LTP should involve overlapping or shared mechanisms capable of producing mutual occlusion. |
| Fusion blockers block LTP. | The mechanism underlying LTP should depend on membrane fusion-related processes sensitive to fusion blockers. |
| LTP stimulation is associated with lateral movement of vesicles containing AMPA receptor subunits (Rumpel et al., 2005; Makino & Malinow, 2009; (Granger et al., 2013) & distribution of these receptors to the membranes (Shi et al., 1999; Passafaro et al., 2001). Blockade of exocytosis of these vesicles cause significant reduction in LTP (Kennedy et al., 2010; Ahmad et al., 2012). Endocytosis of AMPA receptors leads to LTP decay and memory loss (Dong et al., 2015). | Vesicular exocytosis and AMPA receptor trafficking should contribute mechanistically to the induction and maintenance of LTP, while endocytosis should contribute to its reversal or decay. |
| LTP is associated with the enlargement of dendritic spine heads (Lang et al., 2004). | Structural enlargement of dendritic spines should contribute functionally to the induction or maintenance of LTP. |
| Small spines have been identified as preferential sites for the cellular changes associated with LTP induction (Matsuzaki et al., 2004). | Small dendritic spines should possess structural or biochemical properties that make them preferentially susceptible to LTP-associated modifications. |
| High-energy stimulation alone can bypass the requirement of lateral movement of vesicles containing AMPA receptor subunits (Makino & Malinow, 2009) for LTP induction. Instead, LTP can be directly induced even in the absence of AMPA receptor trafficking (Herring & Nicoll, 2016). | The mechanism underlying LTP should permit induction through multiple routes, including pathways that bypass AMPA receptor trafficking under strong stimulation conditions. |
| LTP induction occurs after a delay of at least 20 to 30 seconds (Gustafsson & Wigström, 1990) and over a minute (Escobar et al., 2007), and in some studies, peak reached by only by five minutes (Volianskis et al., 2013). | The mechanism underlying LTP should account for the delayed onset of potentiation following stimulation. |
| LTP induction requires both activation of NMDA receptors by synaptically released glutamate and depolarization of the postsynaptic membrane (Kauer et al., 1988). | The mechanism underlying LTP should depend on coincident glutamatergic activation and postsynaptic depolarization mediated through NMDA receptor activation and calcium influx. |
| In excitatory neurons, spine depolarization can occur without subsequent dendritic depolarization (Beaulieu-Laroche and Harnett, 2018). Moreover, distal dendrites in humans contribute only limited excitation to the soma, even during dendritic spikes (Beaulieu- Laroche et al., 2018). | Learning-related computations should be capable of occurring locally at dendritic spines without requiring action potential generation. |
| b) To explain dendritic spikes, high voltage events recorded on different location of dendrites | |
| A dendritic spike occurs when the summation of approximately 10 to 50 postsynaptic potentials (at the spines) takes place (Antic et al., 2010). | The mechanism underlying dendritic spikes must explain how coordinated activity across multiple spines generates sufficient local depolarization. |
| Some dendritic spikes do not lead to somatic action potentials (Golding & Spruston, 1998). | The mechanism underlying dendritic spikes must account for local dendritic processing that can occur independently of somatic action potential generation. |
| Current injection into the dendrites of human layer 2/3 neurons generates repetitive trains of fast dendritic calcium spikes that occur independently of somatic action potentials (Gidon et al., 2020). | Dendritic electrical events should be capable of remaining spatially restricted and functionally independent from somatic firing. |
| The prevalence of dendritic spikes on the dendrites of place cells (CA1 neurons) in behaving mice is predictive of spatial precision (Sheffield & Dombeck, 2015). | The mechanism underlying dendritic spikes should explain their relationship to spatially precise information processing. |
| Standard model of dendritic spike during natural sensory stimuli (Smith et al., 2013) assumes near-simultaneous activation of dozens of synapses on a single dendritic branch of a neuron. But, in vivo functional imaging studies show that sensory stimuli drive sparse, scattered synaptic activity across the dendritic arbor, not dense branch-specific clusters (Jia et al., 2010; Iacaruso et al., 2017) because axons from a common source make sparse, randomly distributed connections onto a target neuron's dendrites (Kasthuri et al., 2015). Hence, biophysical requirement of dense co-activation of ~40 synapses within a short dendritic segment for generating an NMDA spike is not yet observed under natural sensory driving. | The mechanism underlying dendritic spike generation must account for how strong local dendritic depolarization can arise despite sparse and spatially distributed synaptic activation during natural sensory stimulation. |
| Dendritic spikes a) mediate a stronger form of LTP that necessitates the spatial proximity of associated synaptic inputs (Hardie & Spruston, 2009), b) serve as a mechanism for cooperative LTP (Golding et al., 2002), & c) are essential for single-burst LTP (Remy & Spruston, 2007). One of the requirements for LTP is postsynaptic depolarization, which can result from large EPSPs that trigger dendritic spikes (Hardie & Spruston, 2009). | The mechanism underlying dendritic spikes should explain how coordinated activation across multiple synaptic inputs contributes to postsynaptic depolarization and LTP induction. |
| Domain 3 Associative & Computational | |
| Ability to generate near-infinite number of first-person properties using a finite number of cells and their processes. | The mechanism must support combinatorial operations capable of generating a very large repertoire of internal experiences using a finite number of neurons and synaptic elements. |
| A dynamically adapting circuit mechanism. | The mechanism must support continual incorporation of new learning while also permitting modification, weakening, or reversal of previously established changes. |
| The ability to store new memories without overwriting existing ones. | The mechanism must support formation of new associative memories without catastrophic interference with previously stored information. |
| A framework for a mechanism that enables the system to generate hypotheses (Abbott, 2008). | The mechanism should support indirect associations and inferential processing that allow relationships between previously unassociated items to be derived from overlapping associative structures. |
| Circuits with identical synaptic connectivity can function differently (Marder, 2012). | Functional differences between circuits cannot be explained solely by static synaptic connectivity and therefore require additional dynamic or context-dependent mechanisms. |
| A study (Hedger et al., 2026) demonstrates perceptual integration is achieved through shared representational resources rather than exhaustive, pairwise associations. Vicarious body maps that bridge vision & touch explains a fundamental organizational principle of the brain. | The mechanism should support reuse of shared representational substrates across multiple sensory and contextual associations in a scalable manner. |
| A study (Tafazoli et al. 2026) demonstrates that learning gives rise to shared, task-general neural subspaces that are reused across different behavioral contexts. | The mechanism should support stable and reusable neural subspaces that can be flexibly recruited across different behavioral contexts. |
| Completion of the whole memory during recall using any part of it (Rolls, 2013). Even partial features of one associatively learned item can trigger the memory of the second item. | The mechanism must support pattern completion, enabling partial cues to retrieve complete associative memories. |
| The firing of the same individual neurons in the prefrontal cortex prior to speaking identical phonetic words, such as 'sea' & 'see.' (Khanna et al., 2024). | The mechanism should explain how distinct meanings can generate identical phonetic outputs through shared downstream neuronal activity. |
| Domain 4. Temporal & Dynamical | |
| During memory retrieval, a subset of neurons that were previously unresponsive to the cue stimulus become active (Schlack & Albright, 2007; Furtak et al., 2007). | The mechanism must explain how memory retrieval recruits additional neurons that were previously unresponsive to the cue stimulus. |
| Representational drift" refers to the phenomenon in which the specific set of neurons activated during a repeated brain function gradually changes over time (Schoonover et al., 2021; Marks & Goard, 2021; Deitch et al., 2021). | The mechanism must support stable behavioral or cognitive functions despite gradual changes in the specific neuronal populations participating over time. |
| The system requires a sleep state for approximately one-third of its operating time. In a predator-prey environment, this is an unsafe state. | The mechanism should explain why prolonged periods of reduced behavioral responsiveness are biologically necessary despite substantial evolutionary disadvantages. |
| While living aboard a space station, the need for sleep decreases by more than an hour (Dijk et al., 2001; Gonfalone, 2016). | The mechanism should account for the relationship between sensory/environmental input levels and sleep requirement. |
| When a memory is retrieved, it enters a transiently unstable, or labile state (Judge & Quartermain, 1982). | Retrieval-associated processes should permit temporary modification or updating of stored memories. |
| Firing of lateral amygdala (LA) neurons becomes more synchronized through modulation of theta frequency within the LA (Pare´ & Collins, 2000). Synchronous oscillations in the theta & gamma bands are observed between the basolateral amygdala (BLA) & interconnected brain regions during the retrieval & consolidation of fear memories (Bauer et al.,2004; Seidenbecher et al., 2003). | The mechanism underlying fear memory retrieval should be compatible with synchronized oscillatory activity across interconnected brain regions. |
| The power spectrum of local field potentials (LFPs) has been reported to scale as the inverse of the frequency, but the origin of this 1/f noise is at present unclear (Bédard & Destexhe, 2009). | The operational architecture should explain how neural activity generates scale-free dynamics and 1/f structure in local field potential power spectra. |
| Domain 5. Systems level | |
| Transfer of learning (Dahlin et al., 2008) | The mechanism should support reuse of partially overlapping processing components across different learning contexts. |
| Ability to generalize. | The mechanism must support shared representations that enable generalization across related experiences and retrieval events. |
| Hippocampal neurons fire when an animal reach a specific place. They also fire during different extra-spatial cognitive functions such as motion trajectory (Frank et al., 2000), localization & memory retrieval in different contexts (Pastalkova et al., 2008), response to reward (Gauthier & Tank, 2018), response to auditory frequency in cognitive tasks (Aronov et al., 2017), formation of visual map (Killian et al., 2012), mental navigation (Neupane et al., 2024), organization of conceptual knowledge (Constantinescu et al., 2016), & abstract learning (Schuck & Niv, 2019; Park et al., 2020). Visual images lead to firing of sparsely located hippocampal neurons (Waydo et al., 2006). | The mechanism should explain how individual hippocampal neurons can participate flexibly across diverse spatial, sensory, conceptual, and cognitive functions. |
| Coupled ripple oscillations between the medial temporal lobe and neocortex retrieve human memory (Vaz et al., 2019). | The mechanism underlying memory retrieval should support coordinated integration across medial temporal and neocortical regions during ripple oscillations. |
| Representational drift" refers to the phenomenon in which the specific set of neurons activated during a repeated brain function gradually changes over time (Schoonover et al., 2021; Marks & Goard, 2021; Deitch et al., 2021). | The mechanism must preserve stable cognitive function despite gradual changes in the neuronal populations participating in a given representation. |
| The mechanism utilizes pre-existing schemas (Tse et al., 2007), which are expected to be used interchangeably. | The mechanism should support reuse and flexible integration of pre-existing schemas during new learning. |
| Rapid changes in both the magnitude & correlational structure of cortical network activity (Benisty et al., 2024). | The operational framework should support rapidly reconfigurable functional connectivity. |
| Domain 6. Pathological a) Seizures | |
| The intracellular electrophysiological correlate of epileptiform activity is the paroxysmal depolarizing shift (PDS), characterized as a giant excitatory postsynaptic potential (EPSP) (Johnson & Brown, 1981). | The mechanism must explain how large, synchronized depolarizing events arise during seizures and propagate across neighboring neural regions. |
| Various seizures (particularly when the onset is at the hippocampus) are associated with distinct types of hallucinations. | The mechanism should explain how abnormal neural activity during seizures can generate internally experienced sensory phenomena in the absence of external stimuli. |
| The pathological changes associated with amyotrophic lateral sclerosis (ALS) spread laterally. | The mechanism should account for the progressive lateral spread of pathological changes observed in ALS. |
| In animal models of seizures, the transfer of injected dye from one CA1 neuron to neighboring CA1 neurons has been observed (Colling et al., 1996). | Seizure activity should be capable of increasing abnormal intercellular communication or permeability between neighboring neurons. |
Cell swelling is commonly observed during the "spreading depression" phase of seizures (Kempski et al., 2000; Olsson et al., 2006; Colbourn et al., 2021). | The mechanism should explain how seizure-associated cellular swelling contributes to propagation of pathological electrical activity. |
| Loss of dendritic spines occurs after kindling, during seizures, & following the induction of long-term potentiation (LTP). | The mechanism should explain why conditions associated with excessive neural activation are accompanied by dendritic spine loss. |
| Seizure disorders are often linked to neurodegenerative changes (Farrell et al., 2017). | The mechanism should account for how prolonged or repeated pathological neural activity contributes to progressive neurodegeneration. |
| CA2 region of hippocampus is resistant to seizures, LTP induction, ischemic & hypoxic injuries (Kirino, 1982; Sadowski et al., 1999). | Structural or molecular properties unique to the CA2 region should explain its relative resistance to seizures, LTP induction, ischemia, and hypoxic injury. |
| Loss of consciousness is a common feature during complex seizures. | The mechanism underlying consciousness should be vulnerable to disruption by large-scale pathological synchronization during seizures. |
| Multiple vertical subpial resections have been shown to alleviate seizures (Morrell et al, 1989). | Seizure propagation should depend on laterally organized pathways that can be disrupted by vertical subpial resections. |
| b) Schizhophrenia | |
| Auditory hallucinations are a common symptom of schizophrenia. | The mechanism should explain how internally generated perceptual experiences can occur in the absence of corresponding external sensory input. |
| Schizophrenia is characterized by impaired working memory performance (Goldman-Rakic, 1994). | The mechanism should explain how pathological changes disrupt the stability, specificity, or maintenance of working memory representations. |
| Spontaneous activity of dopaminergic neurons in the ventral tegmental area (VTA) has been linked to the emergence of psychotic symptoms (Liddle et al., 2000; Lodge et al., 2007). Also, hyperactivity of the striatal dopamine system is associated with schizophrenia (Brunelin et al., 2013). | Dopaminergic dysregulation should influence neural mechanisms involved in associative processing, salience assignment, or internally generated perceptual experiences. |
| Dopamine antagonists are a primary class of medications used to treat schizophrenia. | The mechanism should explain why reduction of dopaminergic signaling alleviates psychotic symptoms. |
| Altered consciousness in schizophrenia (Berkovitch et al.,2017). Schizophrenia is characterized by a profound alteration in aspects of consciousness, such as self-relatedness & the ability to relate to the external world (Urfer-Parnas et al., 2010). | The mechanism should account for disturbances in self-related processing and altered relationships between internally generated experiences and external reality. |
| c) Pleasure | |
| Exposure to cocaine results in the attenuation of postsynaptic potentials in the MSN spines of the nucleus accumbens (NAc) (Beurrier & Malenka, 2002). | The mechanism must explain how cocaine-induced dopaminergic modulation of the spines of MSNs of the NAc, leading to attenuation of postsynaptic potentials. |
| In the “addicted” state, there is an impaired ability to induce LTD at the input synaptic regions of MSNs in the nucleus accumbens (NAc) (Kasanetz et al., 2010). | The mechanism must account for the reduced ability to induce LTD at NAc synapses in the addicted state. |
| The inner sensation of pleasure is associated with specific properties of the nucleus accumbens (NAc), as reflected by the ability to induce LTD at the input synapses of its medium spiny neurons (MSNs). | The mechanism must explain how changes in the NAc are functionally linked to the subjective experience of reward or pleasure. |
| d) Headache | |
| Some anti-seizure medications (e.g. topiramate) is an effective in alleviating migraine headaches (Paungarttner et al., 2023; Pearl et al., 2023). | The mechanism underlying migraine should account for its modulation by anti-seizure medications, suggesting shared components of neuronal excitability or network-level propagation with seizure-related processes. |
| Post-ictal headache (Caprara et al., 2020). | The mechanism underlying seizures should account for post-ictal headache as a residual manifestation of seizure-related neural activity. |
| Botulinum toxin, local anesthetic agents, & plastic surgery (Becker, 2020; Robbins et al., 2014; Kung et al., 2011), oxygen (Cohen et al., 2009), dopamine agonists & dopamine antagonists are used in treating different headache pains. | The mechanism underlying headache should be modulated by a diverse set of interventions, including pharmacological, neural, vascular, and peripheral approaches, suggesting a common downstream pathway of pain generation or propagation. |
| Domain 7. Phenomenological | |
| First-person property of perception. | The mechanism underlying memory formation should share core operational principles with those underlying the generation of conscious perceptual experience, while differing in temporal persistence and reversibility. |
| Mechanism that generate memory must have certain shared features with the mechanism that generates first-person property of memory, with the exception that the mechanism is very short-lived and reverses back very quickly. | The mechanism should allow transient, rapidly reversible states underlying conscious experience, in contrast to more stable states supporting memory storage. |
| Phantom limb sensation. | The mechanism underlying conscious experience must account for the generation of sensory qualia in the absence of corresponding physical structures. |
| Innate behaviors, such as the sucking reflex, are hardwired responses present at birth that support survival. | The mechanism must accommodate genetically specified or developmentally constrained circuits that generate stereotyped behavioral responses to specific stimuli. |
| Domain 8. Evolutionary | |
| As cortical neurons migrate from the periventricular region to their final destinations, the diffusion of dye from an injected neuron to neighboring neurons suggests the presence of intercellular fusion pores (Bittman et al., 1997). This phenomenon is observed in all migrating neurons. This stage is followed by the death of approximately 70% of these cells, with only about 30% surviving. Following dye diffusion, a significant loss of neurons (~ 70%) (Blaschke et al., 1996) & spine loss (ranging from 13% to 20%) occur. | The mechanism must explain how transient intercellular permeability during neuronal migration is followed by large-scale selective neuronal survival and programmed cell elimination. |
| Aging is considered the primary risk factor for neurodegenerative disorders, including Alzheimer’s disease (Guerreiro & Bras, 2015). The histological features of amyloid (senile) plaques & neurofibrillary tangles, typically associated with Alzheimer's disease & a range of neurodegenerative disorders, are also observed in normal aging (Anderton, 1997). | The mechanism underlying neurodegenerative disorders must explain why pathological hallmarks such as amyloid plaques and neurofibrillary tangles also appear in normal aging without necessarily producing clinical dysfunction. The mechanism should explain how early developmental states involving transient intercellular connectivity transition into stable, functionally independent neuronal circuits in the mature brain. |