Solution
This work incorporated the following general logic and principles behind the findings in different biological systems.
- Using finite resources to generate infinitely many outputs: When resources are limited and the system has to manage large number of situations using them, then the system uses a combinatorial mechanism. e.g. generation of nearly 1011 specific antibodies from a finite number of variable (V), diversity (D), joining (J), and constant (C) gene segments (Tonegawa, 1983; Janeway et al., 2001). e.g. Heavy chain: V (~38-46), D(23), J(6), C(9). Light chain: V(29-36), J(4-5), C(1-5). Note: Number of genes in brackets.
- Interconnected paths are not always the major paths: Biochemical & neural transmission pathways from one nodal point often have many paths in different directions. Some are highly prominent in contrast to others. e. g. 1) Dopamine has different actions: a) endocrine action, b) enlarges dendritic spines, c) inhibits interneurons via D2 receptors causing disinhibition, 2) The consequence of vesicle exocytosis a) in contributing the vesicle membrane segments to the cell membranes & b) vesicle contents to the cell membranes. Both may be equally important. 3) O2 is used in a) oxidative phosphorylation for ATP synthesis in mitochondria, b) as a reactant in a chemical process, c) its action as an oxidizing agent. Use only function that integrates seamlessly into the broader solution. This selective alignment is the essence of the approach.
- Learning is expected to generate specific signatures within neural circuits: The extracellular matrix (ECM) acts as an insulating medium that prevents leaky connections between neuronal processes, analogous to the insulation between components on a printed circuit board. Regions in which the ECM is very thin relative to the membrane thickness (Fig.1) may permit the selective formation of connections between adjacent neuronal processes during learning. The preservation of the ECM’s insulating properties, together with the capacity to establish specific connections across it, provides features well suited for information storage. Furthermore, the ability of the ECM to reverse or erase these alterations immediately following learning, analogous to wiping a blackboard clean after writing, underscores its functional significance.
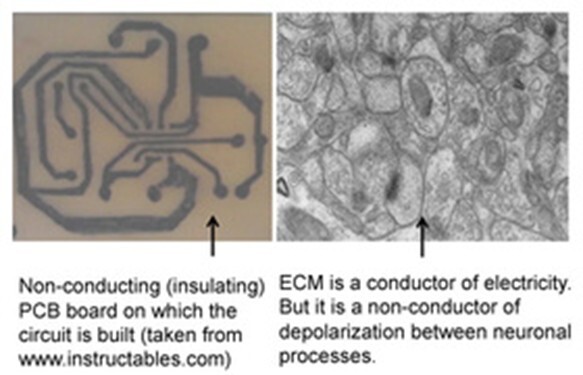
Figure 1. Difference between a printed circuit board (PCB) and the brain. In a PCB, electrical paths that connect electronic components are separated from each other by large area of non-conductive (insulating) material. But in the brain, neuronal processes are separated by very thin (& often negligible) insulating medium of extracellular matrix (ECM). Left side: A printed circuit board made of a non-conducting plate on which conductive tracks are etched to connect the circuit components. Note that the surface area of non-conducting plate that does not have the conductive tracks is roughly more than 80% of the surface area of this plate. Right side: An electron microscopic image from the brain cortex. Note that neuronal & glial cell processes occupy most of the surface area with only very negligible insulating ECM space in between them. Note that while acting as an insulting medium, ECM also has two additional functions. 1) Acts as a buffer zone that facilitates ion flux across membranes. 2) Brain functions occur only in a narrow range of frequency of oscillations of potentials within ECM that spans throughout the cortex. It is to be noted that the negligible ECM has to function very faithfully as an insulating medium without causing spread of depolarization to non-targeted neuronal processes. According to the IPL mechanism, the negligible ECM has an added advantage to etch IPLs between abutted spines. Note that even though it may seem easy for forming an IPL between abutted spines, very high energy is required to displace the hydration water between two lipid membranes (Cohen and Melikyan, 2004; Martens and McMahon, 2008). Furthermore, since the repulsive “hydration force” increases steeply when distance between the two bilayers reduces below 20 Å, fusion between two membranes becomes a very high energy requiring process (Rand and Parsegian, 1984; Harrison, 2015). However, it is anticipated that robust molecular mechanisms are present to overcome this energy barrier.
- Binding: A mechanism that maintains the coherence of structure–function units, a property referred to as binding. Functional coherence of these units requires at least one specific binding feature, and perturbation of this feature leads to measurable alterations in function.
- Fast computation: Since the nature of retrieved memories keeps changing in response to the specific features of the cue stimulus (in sub-second time scales), it is assumed that a fast computation is involved in generating memory.
Suitable brain function to search for the solution → Learning & Memory
Because
- Experiments can be conducted to examine learning-induced changes.
- Cue-induced memory retrieval, experienced as first-person internal sensations on physiological timescales, can be interrupted using specific methods.
- Existing data can be utilized for retrodictive analysis.
Stage I. Finding a solution for behavior
Imagine that two sensory stimuli, stimulus 1 and stimulus 2, undergo associative learning. Later, when stimulus 1 (the cue stimulus) is presented, it is expected to evoke the memory associated with stimulus 2, expressed as behavior that is reminiscent of the presence or arrival of stimulus 2. For this to occur, changes must take place at the convergence points along the pathways through which stimulus 1 and stimulus 2 propagate during the learning process (Fig.2). It is worth noting that the hippocampus, a brain region closely linked to learning and memory, receives inputs from ALL sensory modalities after multiple (approximately 4–8) hierarchical synaptic stages from their respective sensory receptors (Van Hoesen,1982; Lavenex & Amaral, 2000; Rolls, 2007).
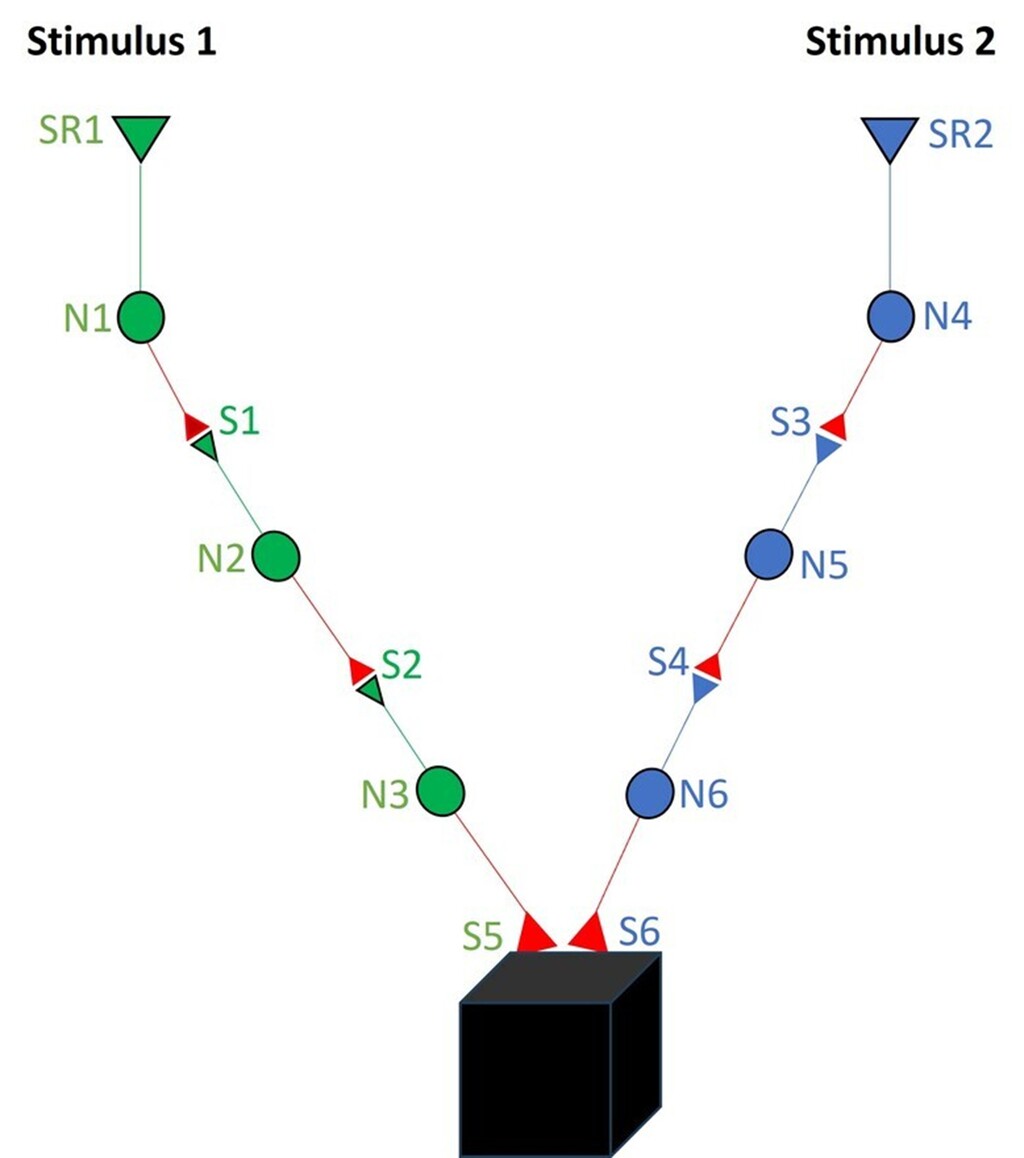
Figure 2. Changes are anticipated to occur at the locations of convergence of associatively learned inputs. Stimuli from all the sensory systems reach the hippocampus after 3 (olfactory) to 8 (auditory) neuronal orders from the respective sensory receptors. For simplicity, this figure has shown only 3 neuronal orders. Neurons from stimulus1 are marked N1, N2 and N3. Neurons from stimulus2 are marked N4, N5 and N6. The junction between the output (presynaptic) terminal of one neuron and the input (postsynaptic) terminal (or dendritic spine) is a synapse. For simplicity, only one each of the input and output terminals are only shown per neuron, although each neuron possesses thousands of such terminals. When associative learning takes place between two sensory stimuli 1 and 2, they activate their corresponding sensory receptors, and the stimulus-induced depolarizations propagate through their synaptically-connected neuronal paths. We want to know the location & the type of change that occurs during learning. It is reasonable to expect that learning cause certain changes at the locations of convergence of Stimuli 1 and 2. After learning, when Stimulus 1 arrives (as a cue stimulus), it should be able to a) generate internal sensation of features of Stimulus 2, which we call as memory of Stimulus 2 in physiological timescales of milliseconds, and b) produce behavioral motor actions reminiscent of Stimulus 2, and c) explain how the learning-change can reverse back for explaining working memory and get stabilized for long-term memory. The still unknown mechanism is shown as a black box next to this synaptic junctions S5 and S6.
Now, let us explore what changes occur at the location of convergence between these two sensory stimuli during learning. What critical changes must occur between the synapses activated by stimulus 1 and stimulus 2? Where specifically should these changes take place in the synaptic network? The interaction should occur between the sub-synaptic locations that enable memory retrieval of the second stimulus when the first stimulus arrives, and vice versa. Examination of electron microscopic images shows that neuronal processes and other cell types are closely apposed, with only a very thin extracellular matrix (ECM) between them (Fig.3). Furthermore, since the mean inter-spine distance is greater than the mean spine diameter (Konur, 2003) (see Fig. 2 on the home page), interactions between subsynaptic locations of different synapses are highly plausible.
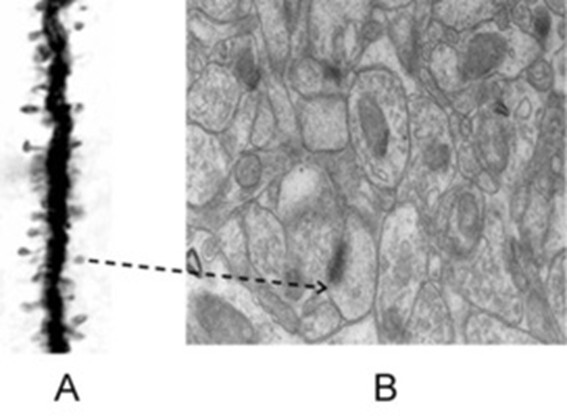
Figure 3. Figures illustrate the importance of inferring that the nearest spine to a given spine on the dendritic branch of a neuron is most likely to belong to a different dendrite, which is probably from another neuron. A) Golgi staining reveals a dendritic branch with spines, which are the inputs to a neuron. The output terminals of the preceding neurons that synapse with these spines are not stained by the Golgi method, suggesting their presence adjacent to the spines. This assumption is supported by electron microscopy, as shown in Figure B, where both the dendritic spines and output terminals can be observed. In this context, nearly any 140 such inputs arriving at the neuronal axon hillock will fire the neuron, leading to the propagation of a signal to all of its output terminals. B) The electron microscopic image demonstrates the crowded nature of neuronal processes and other cells within the neural environment. The extracellular matrix, which exists between neuronal processes and glial cells, serves as an insulating medium that prevents the spread of signals between different neurons that are not connected. Notably, this space is very thin. Arrow: The arrow in Figure A points to a spine, which is further shown in Figure B. The spine in Figure B is marked by a postsynaptic density (PSD), indicated by a dense dark area. Adjacent to the spine is a presynaptic terminal containing synaptic vesicles (for further clarity, refer to Figure 9). As observed in Figure A, the mean inter-spine distance exceeds the mean spine diameter. It is known that the sister branches on a neuron’s dendritic tree avoid overlapping (Grueber & Sagasti, 2010). This suggests that the nearest spine to any given spine on a dendritic branch likely belongs to another dendrite, which, in turn, most likely belongs to a different neuron. If brain function is to arise through spine-to-spine interactions, and if output from a different neuron is required (as indicated by classical conditioning experiments), then the nearest spine to a given spine on a dendritic branch is most probably located on a dendrite of another neuron (No scale bars are provided in the figures).
By systematically examining combinations of different sub-synaptic areas (Pre1-Pre1; Pre1-Post2, Post2-Pre1; Post2-Post2) to match properties that would allow them to generate units of internal sensation through a trial-and-error method, it is possible to reach the interaction between the postsynaptic terminals (dendritic spines or spines or input terminals) of stimulus 1 and stimulus 2 as a possibility (Fig.4A,B). This matches with the finding that depolarization is often limited to the spines (Beaulieu-Laroche & Harnett, 2028) due to high spine neck resistance.
The interaction between these postsynaptic terminals is referred to as the inter-postsynaptic functional LINK (IPL). The term "functional" emphasizes that the formation of the LINK is a function of depolarization of the postsynaptic terminals activated by stimulus 1 and stimulus 2 during associative learning. IPL formation takes place between the lateral margins of the abutted spines due to the special feature that organization of the endocytic machinery takes place mainly at the lateral spine regions (Racz et al., 2004; Makino & Malinow, 2009; Jacob & Weinberg, 2015) that can alter spine surface area at these locations. During memory retrieval, reactivation of the IPL is triggered by the arrival of activity from either stimulus at its corresponding postsynaptic terminal. The term LINK is written in capital letters to signify that it is a critical component of the hypothesis.
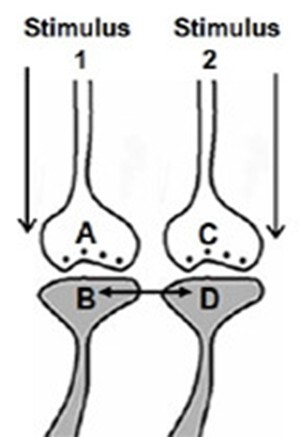
Figure 4A. Illustration depicting the formation of the hypothesized functional LINK between the two postsynaptic membranes B and D during associative learning between stimulus 1 & 2.
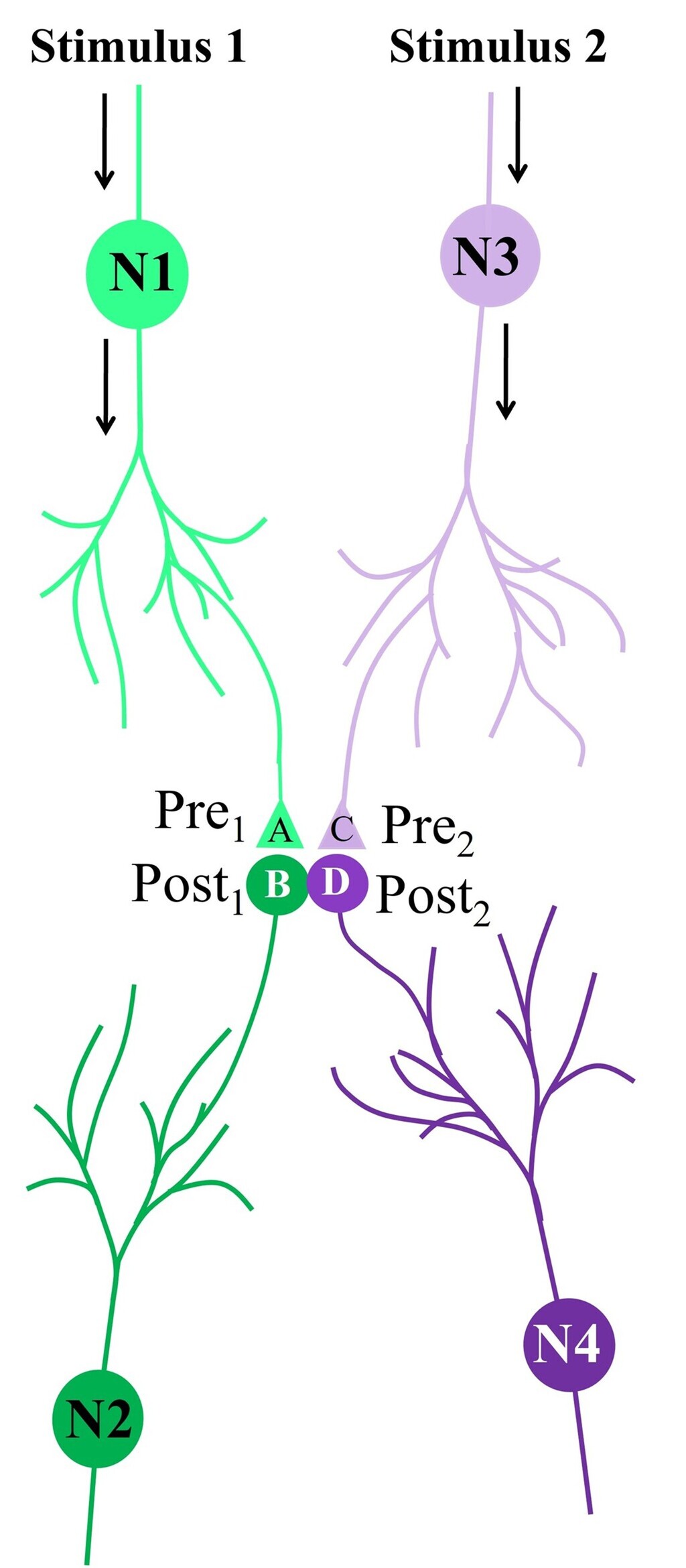
Figure 4B. Illustration of IPL between spines (postsynaptic terminals) B & D that belong to neurons N2 & N4 respectively during associative learning between stimulus 1 & 2.
As the learning events continue, postsynaptic terminals that were previously involved in an earlier learning events will be used to form functional LINKS with a new set of postsynaptic terminals. As learning events progress, it leads to the formation of clusters of inter-LINKed postsynaptic terminals. These clusters, or "islets," are called islets of inter-LINKed spines (IILSPs) (Fig.5).
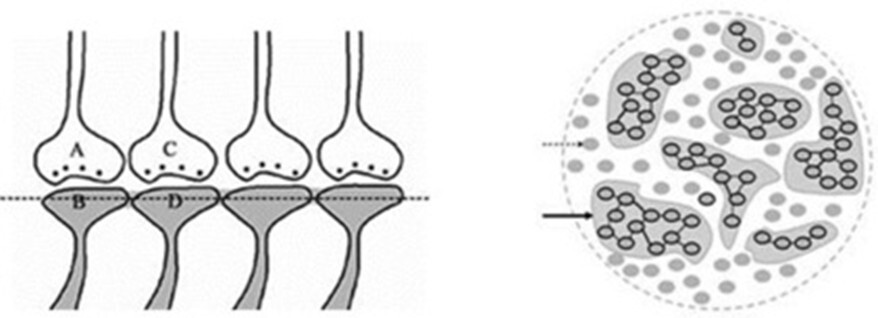
Figure 5. Left Panel: An illustration depicting the formation of islets of LINKed postsynaptic terminals. As learning events continue beyond the initial learning phase, multiple inter-postsynaptic LINKS can form between the involved postsynaptic terminals (dendritic spine heads). In this example, only two presynaptic terminals (A and C) and two postsynaptic terminals (B and D) are marked. Several other postsynaptic terminals are arranged in a horizontal plane. The dotted line represents a cross-section through the inter-LINKed postsynaptic terminals. Right Panel: A hypothetical cross-sectional view of LINKed postsynaptic terminals from synapses in one horizontal plane of a brain region (corresponding to the horizontal dotted line across the postsynaptic membranes in the left panel). In this illustration, all postsynaptic membranes are assumed to be in the same plane. Postsynaptic membranes are represented as small dark circles (broken arrow). During learning, functional LINKs between activated postsynaptic terminals are established. Continued learning involving any of these synapses increases the number of interconnected postsynaptic membranes, forming islets of functionally LINKed postsynaptic terminals (solid arrow). Multiple LINKS within an islet allow for the spread of postsynaptic potentials across the islet. These individual islets are expected to functionally operate independently from one another.
Various types of IPLs are expected to form during associative learning.
Associative learning can take place in sub-second timescales. pdf
Hence, IPL mechanism should be able to take place in this timescale, which can range from nearly hundred milliseconds (see "Unknown" page of this website) to sub-second timescales (see below).
- Removal of hydration layer between postsynaptic terminals: Removal of hydration layer between spines allows them to come in physical contact with each other. This process requires a high amount of energy as demonstrated in experiments using artificial membranes (Rand & Parsegian, 1984; Martens & McMahon, 2008; Harrison, 2015). The IPLs will reverse back quickly. These short-lived IPLs are good candidate mechanisms to explain working memory (Fig.6).
- Partial hemifusion between postsynaptic terminals: A strong interaction between the postsynaptic terminals can lead to a reversible partial hemifusion. This type of interaction explains the retention of learning-induced mechanisms for a longer period compared to the previous type.
- Complete hemifusion between postsynaptic terminals: Further interaction can lead to a reversible complete hemifusion between the postsynaptic terminals. This allows for even longer retention of the learning-induced mechanism.
- Long-term retention through stabilization: If complete hemifusion is maintained for a certain period, it is likely that stabilizing mechanisms will enable the long-term maintenance of this connection, providing lasting changes to the system.
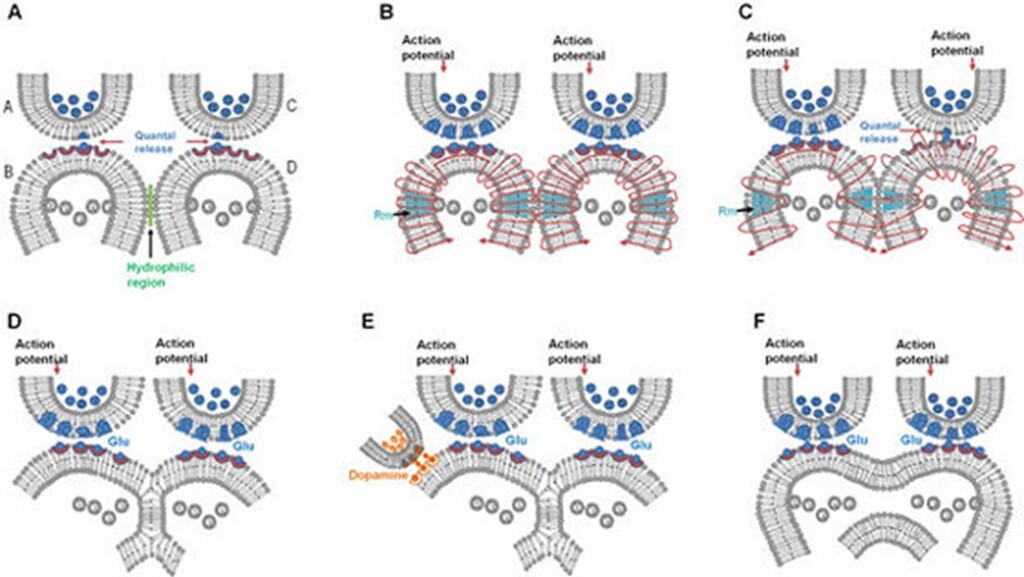
Figure 6. Different types of reversible inter-postsynaptic functional LINKs (IPLs). A) Two abutted synapses A–B and C–D. Presynaptic terminals A and C are shown with synaptic vesicles (in blue color). Postsynaptic terminals (dendritic spines or spines) B and D have membrane-bound vesicles marked V containing subunits of AMPA receptor inside them. Action potential arrives at presynaptic terminal A releasing a volley of neurotransmitters from many synaptic vesicles inducing an excitatory postsynaptic potential (EPSP) at postsynaptic terminal B. From the presynaptic terminal C, one vesicle is shown to release its contents into the synaptic cleft. This quantal release is a continuous process (even during rest) that leads to the generation of very small potentials on postsynaptic membrane D. Note the presence of a hydrophilic region separating postsynaptic terminals B and D. When an action potential arrives at presynaptic terminal A, it activates synapse A–B and generates an EPSP at postsynaptic terminal B. The hydrophilic region prevents any type of interaction between postsynaptic terminals B and D. Very high energy is required for excluding the inter-postsynaptic hydrophilic region (Martens and McMahon 2008). B) Membrane expansion occurring at physiological timescales can provide sufficient energy to exclude the inter-postsynaptic hydrophilic region, allowing close contact between the postsynaptic membranes in this region. This forms a transient inter-postsynaptic LINK that lasts only for a short period of time. During this short period of time, a cue stimulus-generated action potential arriving at synapse A–B reactivates this IPL and spreads to postsynaptic terminal D and induces units of internal sensation at the inter-LINKed postsynaptic terminal D. This can explain working memory. C) Diagram showing formation of a partial inter-postsynaptic membrane hemifusion. These vesicles contain glutamate receptor subtype 1 (GluA1). Activity arriving at the synapse can lead to exocytosis of vesicles containing AMPA GluA1 receptor-subunits abutted to the cell membranes and expansion of the postsynaptic membrane at physiological timescales. During exocytosis, the vesicle membrane gets incorporated into the postsynaptic membrane at locations of exocytosis making this region of the membrane highly re-organizable. This matches with the location where AMPA receptor subunits were shown to concentrate at the extra-synaptic locations extending up to 25nm beyond the synaptic specialization (Jacob and Weinberg 2014). Note the interaction between the outer layers of membranes of the postsynaptic terminals. Depending on the lipid membrane composition, the process of close contact between the membranes described in the above section B) can get converted to a partial hemifusion state. D) Stage of partial hemifusion can progress to complete hemifusion. The reversible partial and complete hemifusions are short-lived and can explain the necessary learning-induced changes responsible for short-term memory. Some of the hemifusion changes can get stabilized for different lengths of time. For example, insertion of a trans-membrane protein across the hemifused segment can maintain the IPL until this protein gets removed. These changes can be responsible for long-term memory. E) Dopamine is known to facilitate motivation-promoted learning. In this diagram dopaminergic input to postsynaptic terminal B that results in latter's expansion, which will augment IPL formation. This can explain the action of dopamine on learning. Furthermore, it can sustain the hemifused LINK for a long period of time, which may facilitate its stabilization. F) Hemifusion can advance to a complete fusion state in pathological conditions and it depends on several factors. Fusion of the postsynaptic terminals between two different neurons can lead to cytoplasmic content mixing and cytotoxic cell response. These include dendritic spine loss and eventually triggering of apoptosis leading to neurodegenerative changes. Note that excessive dopamine can lead to excessive expansion of the postsynaptic membrane and can lead to membrane fusion if other factors that resist this get compromised. Rm: membrane segment marked in Turkish blue shows area where membrane reorganization occurs (Figure modified from Vadakkan, 2015a, b).
Solution to the black box problem
On the Home page, we discussed the black box problem that must be addressed to explain how behavioral motor actions associated with two stimuli learned in association can manifest during memory retrieval (Figs. 1 and 2 on the Home page). The formation of an inter-postsynaptic functional link (IPL) between spines belonging to two different neurons provides a potential solution (Fig.7). This framework also motivates further investigation into a hidden mechanism at or near the IPL that can generate first-person properties reminiscent of the arrival of the memorized item.
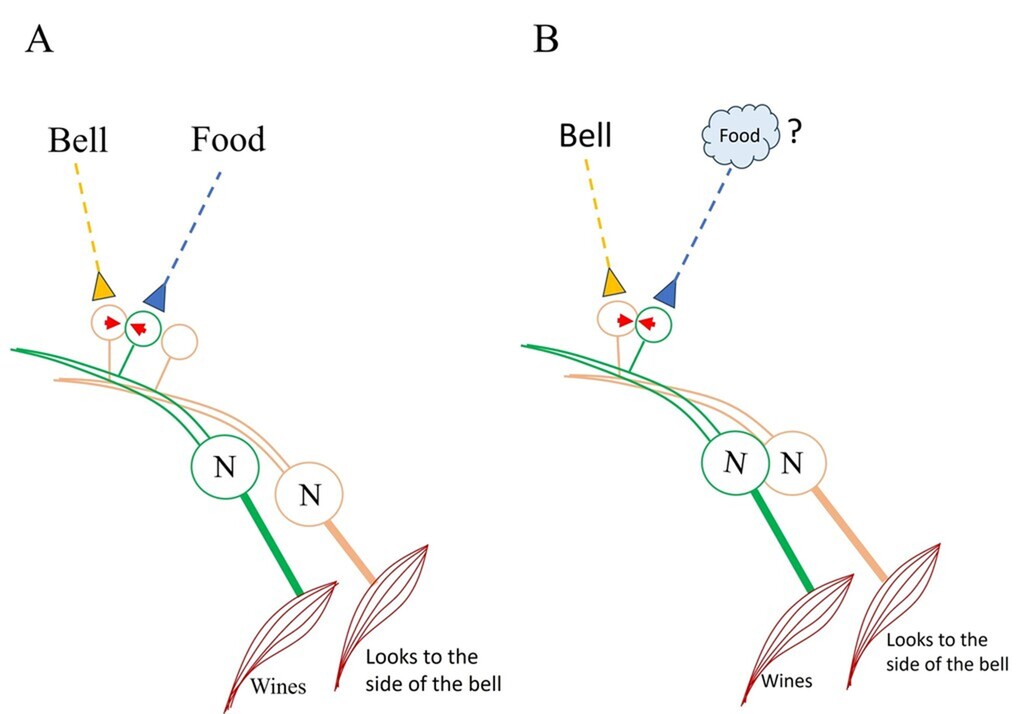
Figure 7. Figure illustrating the solution based on the semblance hypothesis. A: The fact that a) sister branches on a neuron’s dendritic tree avoid overlapping (Grueber & Sagasti (2010) Cold Spring Harb Perspect Biol. 2(9):a001750), and b) the mean inter-spine distance exceeds the average spine diameter (Konur et al., 2003) increases the likelihood that neuronal processes from different neurons – as well as non-neuronal cell processes – are present in the intervening space. When stimuli from the bell and food simultaneously arrive at adjacent (abutting) spines of different dendrites – typically from different neurons – an interaction between these spines can occur. This interaction facilitates the formation of a functional LINK during learning (indicated by red arrowheads). B. Following learning, the presentation of the bell alone can trigger depolarization at the spine that was previously activated by the food stimulus. This leads to: a) behavioral and secretomotor responses such as whining, which reflect the retrieval of the memory of food, and b) the induction of a semblance (a unit of internal sensation) at the inter-LINKed spine that was part of the original food-stimulus pathway.
Stage II. Examining how first-person properties may be generated in the vicinity of the IPL region
The inquiry began with the question: “Can activation of a postsynaptic terminal, in the absence of an arriving action potential at its corresponding presynaptic terminal, lead to a virtual sensation (i.e., an internal sensation occurring without a sensory stimulus)?” This scenario is unlikely unless the system’s operations are endowed with specific structural and functional features. Two key properties enable IPLs to generate first-person phenomena.
Property I: Lateral activation and dominant baseline state
Since first-person internal sensations are virtual, specific properties at and/or around the IPLs were examined in the search for a unique, simple, and universal mechanism. When the artificial intelligence (AI) community investigated mechanisms of memory in biological systems, one of the founding directors of MIT’s AI Laboratory, Marvin Minsky, hypothesized that memories are cue-specific, cue-induced internal sensations–experiences of a stimulus in its absence (Minsky, 1980). This implies that when Stimulus 1 arrives, it can generate an internal sensation corresponding to Stimulus 2. Since IPLs can generate behavioral motor actions reminiscent of Stimulus 2, the following question arises: “Is there any structural or functional feature at or around the IPL capable of generating postsynaptic activation that mimics presynaptic input, producing an internal sensation of a stimulus?”
At the synaptic level, the presynaptic terminal C continuously releases neurotransmitter molecules in a quantal fashion from its synaptic vesicles into the synaptic cleft, even during sleep. This ongoing release generates miniature excitatory postsynaptic potentials (mEPSPs, or “minis”) at postsynaptic terminal D. Occasionally, when an action potential arrives at the presynaptic terminal, it leads to the generation of a large postsynaptic potential. The ongoing quantal release establishes a dominant baseline state at the synapse: under this condition, any postsynaptic depolarization is normally interpreted as originating from presynaptic input. In such a dominant state, lateral depolarization via an IPL activates the postsynaptic terminal in a manner indistinguishable from presynaptic input. This produces an internal sensation in the absence of external stimulation, effectively creating a cellular-level correlate of first-person experience.
To illustrate the principle, consider an analogy from daily life. A pickpocket exploits a dominant sensory state to mask a perturbation: the continuous, natural movement of the gluteal muscles during walking creates a background sensory pattern. As a result, when the wallet is removed, the brain interprets the sensation as part of expected movement–experiencing it as normal, routine input. A modern demonstration (Video) shows that pickpockets are particularly successful when the victim is walking up or down stairs, during which gluteal muscles are more active and clothing movement is increased. In these conditions, the system’s baseline activity is heightened, masking the new input.
The analogy highlights the general principle: maintaining a dominant baseline activity makes the system susceptible to interpreting lateral or unusual inputs as expected, creating internal sensations without direct external cause. In contrast to the pickpocket example, where no new internal activation is generated, IPL-mediated lateral depolarization actively induces postsynaptic responses that mimic presynaptic input. Since no known toxins can fully block quantal release, this property appears to be a reliably preserved feature of the nervous system.
Note: The fact that mEPSPs cannot be completely blocked, even under experimental conditions, suggests that this is a highly conserved, default function of the nervous system.
Property II: Integration via oscillatory activity
A second systemic requirement for generating inner sensations is network-level integration, which is facilitated by oscillatory activity. A study (Beauchamp et al., 2012) found that electrical stimulation of the visual cortex produces a visual percept (phosphene) only when high-frequency gamma oscillations are induced in the temporo-parietal junction. Oscillations provide a temporal framework that coordinates the activation of multiple IPLs, allowing their lateral depolarizations to be integrated across a network.
In this model, the horizontal component of oscillations corresponds to lateral IPL-mediated spread, while the vertical component arises from synaptic interactions along cortical columns. Inter-postsynaptic activation, which occurs perpendicular to classical trans-synaptic flow, contributes to the observed waveform of cortical oscillations. Maintaining oscillations of extracellular potential within a narrow frequency range is necessary for the normal brain functions (Rusalova, 2006; Bagherzadeh et al., 2020; Engel and Singer, 2021). It can be argued that this ensures that IPL activations are synchronized, enabling the system to generate coherent first-person sensations.
In other words,
First-person properties emerge as a system-level property arising from the interaction of:
- 1. Property I: Lateral IPL-mediated activation of postsynaptic terminals under a dominant baseline state.
- 2. Property II: Integration of multiple IPL activations through oscillatory network dynamics.
Together, these properties enable the nervous system to generate internal sensations that mimic external input, forming the basic building blocks of first-person experience.
Why does this system need sleep?
The explanation cannot be trivial or framed merely as an indispensable requirement; it must reflect a substantive property of system operation. A useful analogy can be drawn from veterinary practice. A common technique for administering injections involves patting and then repeatedly tapping the intended injection site with moderate intensity. After several repetitions–e.g., ten taps–this stimulation becomes the dominant sensory input at that location. When the needle is inserted on the subsequent contact, the animal is less likely to detect it, as the nervous system interprets the sensation as another tap (Video).
It is important to note that if the intensity of the final tap were matched precisely to that of the needle insertion, detection would likely be further reduced. The key principle is that misinterpretation of an unusual stimulus as a routine one requires prior establishment of a dominant sensory background state. Within this framework, the injection is perceived not as a distinct event but as a continuation of ongoing input. To administer a second injection at the same site, the tapping sequence must be repeated to re-establish this dominant baseline. This effectively resets the sensory background, ensuring that the second needle insertion is again interpreted as routine input. While not a perfect analogy, this captures an essential feature relevant to sleep.
Sleep can be viewed as a system-level reset that restores the dominant background state in which postsynaptic depolarizations are primarily driven by presynaptic input originating from the environment. Across evolutionary time, under conditions shaped by circadian cycles, sleep provided a critical window for re-establishing this baseline. During sleep, continuous quantal neurotransmitter release produces ongoing depolarization of spine heads, stabilizing the system into a dominant state. This process underscores the substantive–rather than merely indispensable–role of sleep in maintaining proper nervous system function.
Estimating the qualia of inner sensations
A cue stimulus (e.g. stimulus1) reactivates the IPL and depolarizes the inter-LINKed spine from laterally (Fig.8).
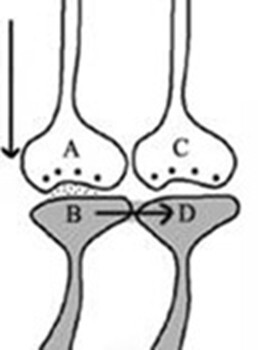
Figure 8. Cue stimulus-induced postsynaptic potential at synapse A-B propagates across the IPL B-D to the inter-LINKed spined D. During retrieval, the cue stimulus activates presynaptic terminal A, leading to the depolarization of its corresponding postsynaptic membrane B. This depolarization is proposed to spread to another postsynaptic membrane, D. Such a spread is possible only if a functional LINK exists between postsynaptic terminals B and D. Consequently, it can be inferred that a functional LINK must be established between these postsynaptic terminals during the learning process.
To characterize the qualia, the subjective sensory qualities, of the inner sensations generated at an inter-LINKed spine when it is depolarized laterally via the IPL, it is necessary to identify the sensory stimuli that can normally depolarize that inter-LINKed spine. To achieve this, one must identify the sensory receptors whose activation can lead to such depolarization. Identifying this set of sensory receptors requires extrapolation from the inter-LINKed spine back to the level of the sensory receptors. Once the full set of sensory receptors is identified, it may become possible to determine many subsets from the set of identified sensory receptors whose activation can depolarize the inter-LINKed spine under examination. A given inter-LINKed spine may be associated with multiple such subsets. The minimal sensory stimulus required to activate one of the identified subsets of sensory receptors that can depolarize the inter-LINKed spine under examination can be considered the basic unit of inner sensation and is termed a “semblion” (Fig.9).
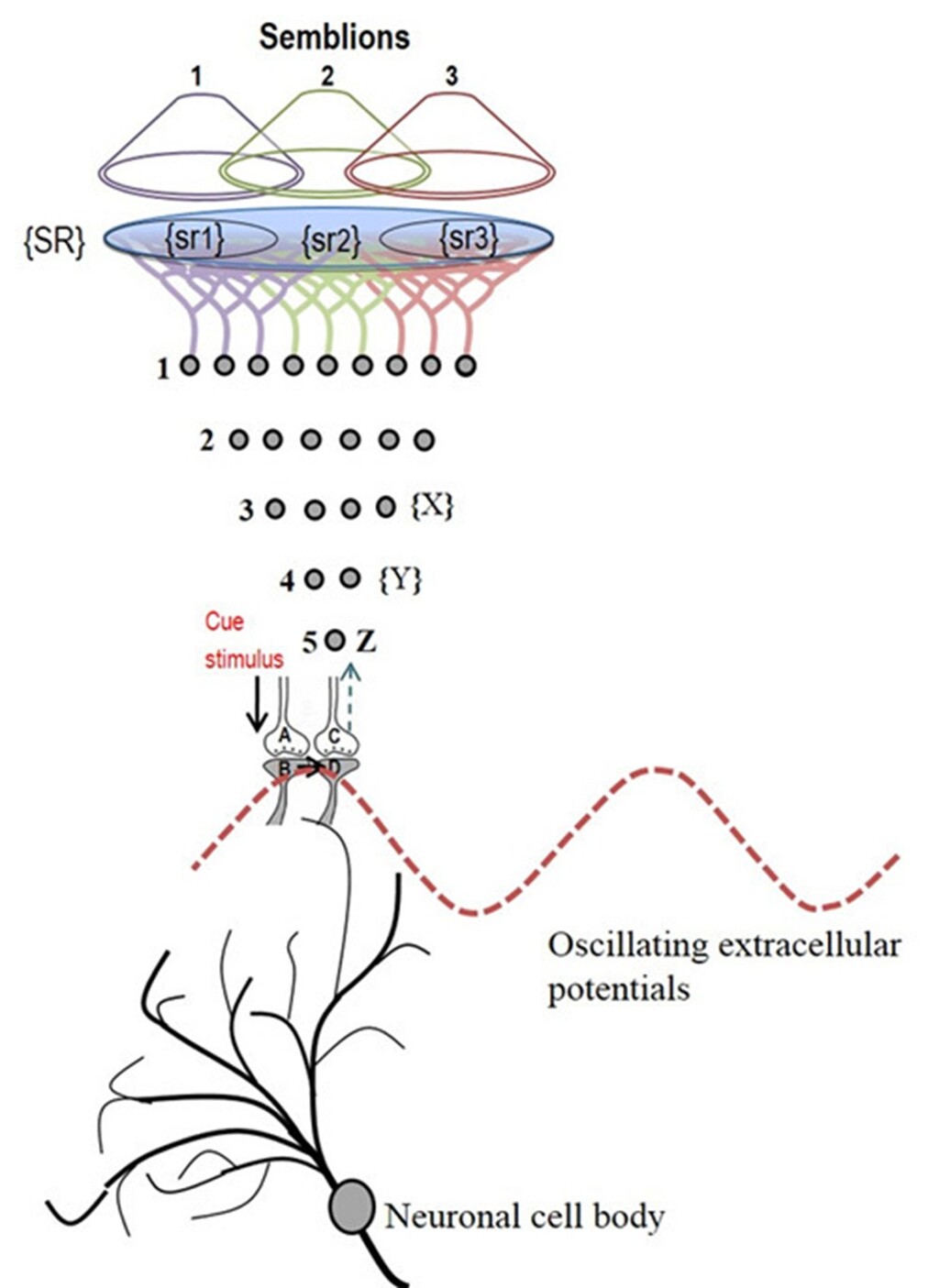
Figure 9. Schematic representation of sensory content of first-person property during reactivation of an inter-postsynaptic functional LINK (IPL). The grey circles represent neurons. The numbers on the left side of the neuronal orders denote their position in relation to the sensory receptors. Neuron Z is shown in neuronal order 5. During memory retrieval, a cue-stimulus reaching presynaptic terminal A depolarizes its postsynaptic membrane B. The resulting excitatory postsynaptic potential (EPSP) re-activates the IPL that activates postsynaptic membrane D from its side, evoking a hallucination that the latter is being depolarized by its presynaptic terminal C. This generates a semblance of arrival of a sensory stimulus from the environment at the presynaptic terminal C. This can be viewed as the basic operational mechanism for the first-person inner sensation. An extrapolation from presynaptic terminal C can be carried out as follows. Presynaptic terminal C belongs to the neuron Z that in turn receives inputs from the set of neurons {Y}. The set of neurons {Y} are activated by the activation of the set of neurons {X}. The set of neurons {X} in turn are activated by the set of neurons in the neuronal order above it. (Recurrent collaterals and projection neurons can also activate a higher order neuron. For simplicity, these are not shown). Continuing this extrapolation towards the sensory level identifies a set of sensory receptors {SR}. It may not be necessary to stimulate the entire receptor set {SR} to stimulate the neuron Z. Stimulation of either one of the subsets of sensory receptor sets {sr1}, {sr2} or {sr3} of the set {SR} may be sufficient to independently activate neuron Z. The dimensions of hypothetical packets of sensory stimuli capable of activating the sensory receptor subsets {sr1}, {sr2} and {sr3} are called semblions 1, 2 and 3 respectively. These semblions are viewed as the basic building blocks of the virtual internal sensations of memory. In the figure, a cue stimulus can cause postsynaptic terminal D to hallucinate about any of the semblions 1, 2, 3 or an integral of them. Since the cue stimulus can reactivate large number of IPLs, it can generate large number of semblions. There is a computation occurring between the semblions that provide the final qualia of inner sensation evoked in response to a specific cue stimulus. Memory is generated only when the brain operates in a narrow range of frequency of oscillating extracellular potentials. Note that synaptic transmission and propagation of potentials across the IPLs in near perpendicular directions contribute vector components to generate the oscillating extracellular potentials (marked by the waveform) (Modified from Vadakkan, 2011).
Computational algorithm expected in the system
A cue stimulus can reactivate large number of IPLs along its path (Fig.10).
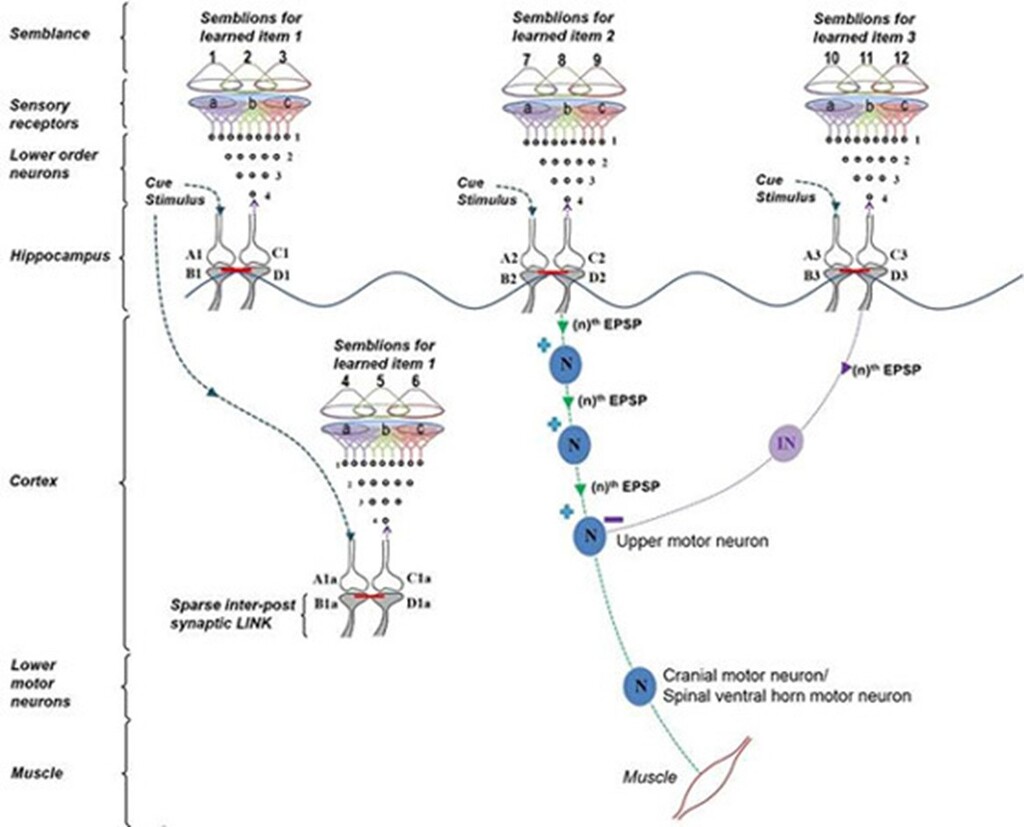
Figure 10. A cue reactivates several IPLs to generate units of inner sensations and elicit motor actions. Oscillating neuronal activity results in the activation of many downstream neurons. They can be kept tonically inhibited under resting conditions (not shown) to subthreshold levels. A cue stimulus undergoes associative learning with item 1 and item 2. Following this, the cue stimulus retrieves memories of items 1 and 2. Along with retrieving memory item 2, the cue stimulus also evokes a motor response through the motor neuron. Later, the same cue stimulus undergoes a second associative learning with item 3. Following this, the cue stimulus evokes internal sensations (semblances) of learned items 1, 2 and 3. However, as the semblance for item 3 was evoked, it results in the inhibition of the motor activity. This is an example of behavioral inhibition in the frontal cortex. Reward-induced associative learning may be facilitated by dopamine-induced enlargement of dendritic spines (Yagishita et al., 2014) that favors IPL formation and stabilization. In the hippocampus, reactivation of certain IPL in response to specific locations can lead to firing of certain subthreshold-activated CA1 neurons (place cells). This explains how spatial memories are associated with place cell firing. EPSP: excitatory postsynaptic potential. nth EPSP: the last EPSP necessary to achieve threshold EPSP to generate an action potential. Each motor action will evoke certain sensory stimulus in the form of proprioception that will act as a feedback stimulus to the system confirming that the motor action was executed. N: Excitatory neuron; IN: Inhibitory neuron. A and C: Presynaptic terminals; B and D: Postsynaptic terminals. Red line between B and D: Inter-postsynaptic LINK. (+) stimulation; (-) inhibition (Modified from Vadakkan, 2015b).
If all the possible semblions that can be elicited at an inter-LINKed spine can be estimated, it becomes possible to deduce the computational algorithm that links these semblions (inputs) to the features of the memorized item (output). This can be illustrated with the following example: when one is asked to recall a rose, multiple features are evoked, including its color, fragrance, texture, thorns, green sepals, and even the taste of its petals. The cue stimulus, the question about the rose, reactivates a specific set of IPLs, inducing semblions at the inter-LINKed spines. These represent the elemental units of inner sensations. Their integration gives rise to the emergent net semblance, or holistic memory representation, of the rose (Fig.11).
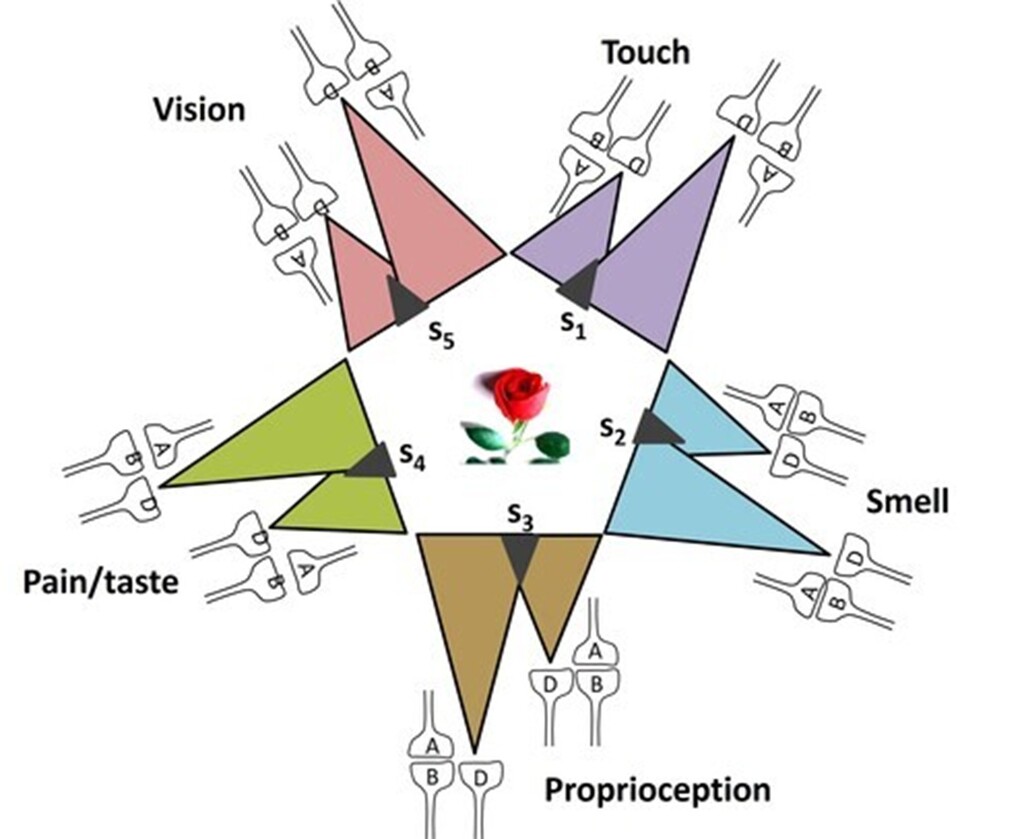
Figure 11. Integration of units of inner sensations from several inter-LINKed spines (marked D). Each IPL encodes different sensory properties with the name "rose flower. A computational algorithm from the unitary semblions to generate the rose can be discovered. Each colored triangle represents retrograde extrapolations from the inter-LINKed spine D to generate semblions. Oscillating extracellular potentials is expected to be responsible for integrating the semblions to generate the memory.
Comparing the integrated products of semblions with the actual sensory features of the retrieved item can provide critical insights into the neural algorithm underlying memory retrieval. Notably, the net semblance can surpass a certain threshold without compromising retrieval accuracy. Because the number of inter-LINKed spines is continuously modified through associative learning over the course of life, the characteristics of the corresponding semblions are also expected to change gradually. This leads to a progressive refinement of net semblances underlying memory.
Brain operation requires maintenance of oscillating extracellular potentials within a narrow frequency range. Differential electrodes are used to measure differences in net extracellular potentials be-tween two brain regions. Such recordings can be obtained intracranially by inserting electrodes directly into brain tissue, typically positioning the electrode tips within the extracellular matrix (ECM). Alternatively, extracellular activity can be recorded from the surface of the dura mater during neurosurgical procedures, or non-invasively from the scalp using electroencephalography (EEG). Across these approaches, it has consistently been observed that normal brain function depends on maintaining oscillating extracellular potentials with-in a relatively narrow frequency range (Rusalova, 2006; Bagherzadeh et al., 2020; Engel and Singer, 2021; Niedermeyer & da Silva, 2017). Deviations from this range, as reflected in EEG waveforms, are closely associated with alterations in the level of consciousness. This principle is applied clinically in monitoring conscious states during the management of conditions such as seizure disorders and encephalopathies.
Neurons maintain a resting membrane potential of approximately −70 mV, representing intracellular voltage relative to the extracellular space. This potential continuously changes as depolarization propagates along the neuronal membrane. Extracellular voltage, measured relative to the intracellular environment, reflects these membrane dynamics. At any given location, extracellular voltage fluctuates in response to changes in intracellular potentials of: a) excitatory neuronal processes in the surrounding region, b) inhibitory neurons inter-connected via gap junctions, and c) calcium wave propagation across astrocytes.
When oscillations of extracellular potentials occur within a system, they can be interpreted as arising from the superposition of propagating electrical activities in multiple directions. Synaptic transmission and propagation of potentials across IPLs (Fig.12), which often occur along near-perpendicular trajectories, contribute distinct vector components. The integration of these components gives rise to the observed oscillatory waveforms. Within this framework, normal brain function can be understood as dependent on maintaining these oscillations within a constrained frequency range.
This constraint has further implications. It suggests that semblance formation, which under-lies the first-person experiential property of the system, emerges as a system-level feature of networks capable of generating and sustaining such coordinated oscillatory activity.
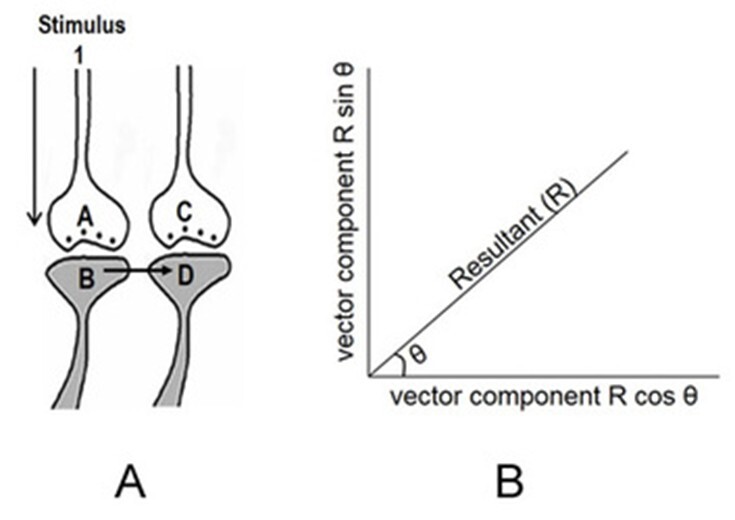
Figure 11. Integration of units of inner sensations from several inter-LINKed spines (marked D). Each IPL encodes different sensory properties with the name "rose flower. A computational algorithm from the unitary semblions to generate the rose can be discovered. Each colored triangle represents retrograde extrapolations from the inter-LINKed spine D to generate semblions. Oscillating extracellular potentials is expected to be responsible for integrating the semblions to generate the memory.
Perception and consciousness
It is reasonable to assume that the mechanism responsible for generating these internal sensations shares core features with the first-person properties underlying other higher brain functions, such as perception and consciousness. Slight modifications in the induction process are likely to give rise to the internal sensations experienced during perception. For perception, the mechanism must account for real-time induction of internal sensations in a manner that aligns with the wide range of known perceptual phenomena. Moreover, IPLs must reverse within milliseconds. This framework has been applied specifically to explain visual perception (Vadakkan, 2015) (Fig.13).
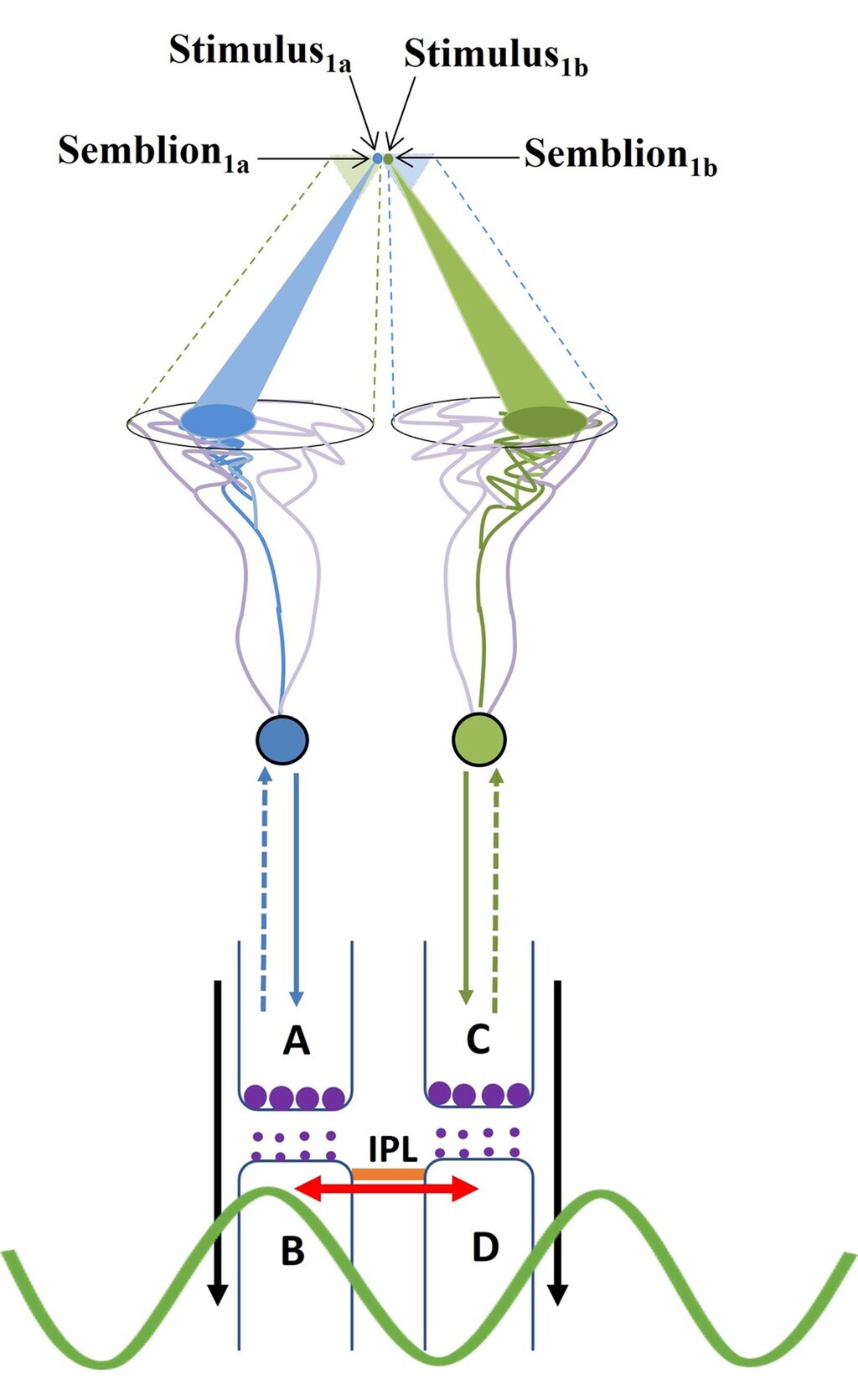
Figure 13. Diagram showing a mechanism for the formation of basic unit of perception. Stimulus1 and Stimulus2 arriving from infinitesimally close locations from an object simultaneously activate the inter postsynaptic functional LINK from the opposite sides resulting in the simultaneous formation of Semblion1 and Semblion2 respectively. Stimulus1 activates the sensory receptor set {srp1} and this activity results in the activation of neuron N1 resulting in activation of the synapse A–B. This results in the activation of the inter-postsynaptic functional LINK B–D evoking a unit of semblance labeled Semblion1 from the receptor set {sr1}. Similarly, Stimulus2 activates the sensory receptor set {srp2} and activity results in activation of the neuron N2 resulting in the activation of the synapse C–D. This results in the activation of the inter-postsynaptic functional LINK B-D evoking a unit of semblance labeled Semblion2 from the receptor set {sr2}. Note that the semblance induced by one stimulus matches the other stimulus. This simultaneous formation of overlapping stimulus-semblance U-loops in opposite directions across the inter-postsynaptic func tional LINK B-D is hypothesized to form the basic units of perception. Interruption of the formation of stimulus-semblance U-loops due to lack of sensory stimulus beyond the edges breaks the continuity in the formation of percept from the surface of the object. Simul taneous formation of stimulus-semblion U-loops arriving from a different plane provides percept of the scenery beyond the edges of the object. The neuronal paths through which stimulus reach the inter-postsynaptic functional LINK is given in blue and green. The neuronal paths through which extrapolation from the postsynapses up to the sensory receptors is carried out to estimate the semblance also include neurons and sensory receptors that are given in violet color. The directions of the stimuli and semblances are opposite to each other. Note that for simplicity, the semblions are drawn to a sin gle point in contrast to their expected spanning over several sensory receptors as in the original derivation (Vadakkan 2013) and shown in Fig. 3 above. The overlapping of perceptons with that of the origin of sensory stimuli from the object occurs only for a limited distance from the eye. w Waveform in front of the inter-postsynaptic junction represents the oscillating surface- or extracellular-recorded potentials whose horizontal component has contributions from the spread of activity through the inter-postsynaptic functional LINK B–D.
In the case of consciousness, the model answers two key questions.
- a) Why are numerous units of internal sensations induced even when the organism is at rest? Continuous oscillating extracellular potentials reflect reactivation of a large number of IPLs. The net semblance generated constitutes the first-person experience of the self at rest.
- b) Is there any benefit in integrating these inner sensations into a single non-specific inner sensation? The majority of sensory inputs arriving at the nervous system are neither beneficial nor deleterious to the organism. Hence, integrating the semblances induced by these in-puts into a neutral baseline state provides an advantage, enabling the system to generate distinct inner sensations in response to both deleterious and beneficial sensory inputs (Vadakkan, 2010; 2015).
Unified mechanism underlying memory, perception, and consciousness
Memory, perception, and consciousness can be understood as different operational expressions of a common underlying mechanism based on IPL-mediated induction and integration of internal sensations.
- In memory retrieval, cue-induced reactivation of specific IPLs generates discrete units of internal sensation, whose integration reconstructs the features of a previously experienced item.
- In perception, ongoing sensory inputs continuously activate IPLs in real time, producing rapidly updating internal sensations that correspond to the external environment. This requires fast reversibility of IPLs to allow moment-to-moment changes.
- In consciousness, large-scale, ongoing reactivation of IPLs, reflected in oscillatory extracellular activity, generates a continuous background of integrated internal sensations. This forms the baseline first-person experience of the self.
Thus, these three functions differ primarily in the source, timing, and extent of IPL activation, while relying on the same fundamental process of generating and integrating internal sensations.
References
Cohen FS, Melikyan GB (2004) The energetics of membrane fusion from binding, through hemifusion, pore formation, and pore enlargement. J. Membr. Biol. 199: 1–14. PubMed
Grueber WB, Sagasti A (2010) Self-avoidance and tiling: Mechanisms of dendrite and axon spacing. Cold Spring Harb Perspect Biol. 2010 Sep;2(9):a001750. PubMed
Harrison SC. (2015) Viral membrane fusion. Virology 479-480:498–507. PubMed
Jacob AL, Weinberg RJ (2014) The organization of AMPA receptor subunits at the postsynaptic membrane. Hippocampus. 25(7):798-812. PubMed
Martens S, McMahon HT (2008) Mechanisms of membrane fusion: disparate players and common principles. Nat Rev Mol Cell Biol 9(7):543–556. PubMed
Rand RP, Parsegian VA (1984) Physical force considerations in model and biological membranes. Can J Biochem Cell Biol 62(8):752–759. PubMed
Vadakkan KI (2007) Semblance of activity at the shared post-synapses and extracellular matrices - A structure function hypothesis of memory. ISBN:978-0-5954-7002-0 Download
Vadakkan KI (2010) Framework of consciousness from semblance of activity at functionally LINKed postsynaptic membranes. Front Psychol. 1:168. Article
Vadakkan KI (2011) Processing semblances induced through inter-postsynaptic functional LINKs, presumed biological parallels of K-lines proposed for building artificial intelligence. Frontiers in Neuroengineering 4:8. PubMed
Vadakkan KI (2012a) The nature of "internal sensations" of higher brain functions may be derived from the design rules for artificial machines that can produce them. Journal of Biological Engineering. 5;6(1):21. PubMed
Vadakkan KI (2013) A supplementary circuit rule-set for the neuronal wiring. Front Hum Neurosci. 7:170. Article
Vadakkan KI (2015a) A framework for the first-person internal sensation of visual perception in mammals and a comparable circuitry for olfactory perception in Drosophila. Springerplus. 4:833. Article
Vadakkan KI (2015b) A pressure-reversible cellular mechanism of general anesthetics capable of altering a possible mechanism for consciousness. SpringerPlus 4:485 PubMed
Vadakkan KI (2016) The functional role of all postsynaptic potentials examined from a first-person frame of reference. Reviews in the Neurosciences (Explained how a neuronal soma is flanked by a large number of internal sensory processing units and their relationship with neuronal firing). Article
Vadakkan KI (2019) From cells to sensations: A window to the physics of mind. Phys Life Rev. 31:44-78. PubMed